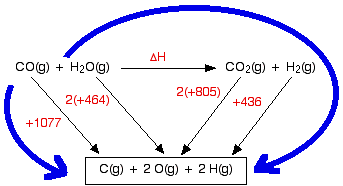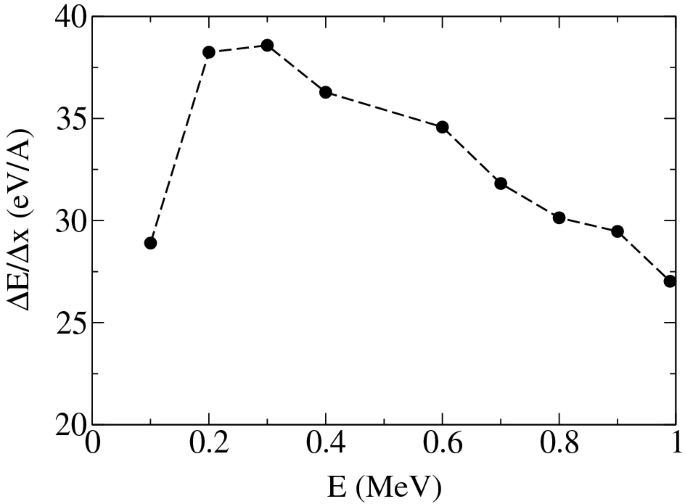
Towards the ionizing radiation induced bond dissociation mechanism in oxygen, water, guanine and DNA fragmentation: a density functional theory simulation | Scientific Reports

Density Functional Theory plus U Study of Methanol Adsorption and Decomposition on CuO Surfaces with Oxygen Vacancy - Aerosol and Air Quality Research

Calculating Bond Enthalpies - Measuring Bond Dissociation Energies with Computational Chemistry - YouTube
![PDF] Oxygen–oxygen bond dissociation enthalpies of di-tert-butyl peroxide and di-trifluoromethyl peroxide | Semantic Scholar PDF] Oxygen–oxygen bond dissociation enthalpies of di-tert-butyl peroxide and di-trifluoromethyl peroxide | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/23ffbaa946efe59c6cdf14cf914b71ab68ba5108/3-Table1-1.png)
PDF] Oxygen–oxygen bond dissociation enthalpies of di-tert-butyl peroxide and di-trifluoromethyl peroxide | Semantic Scholar
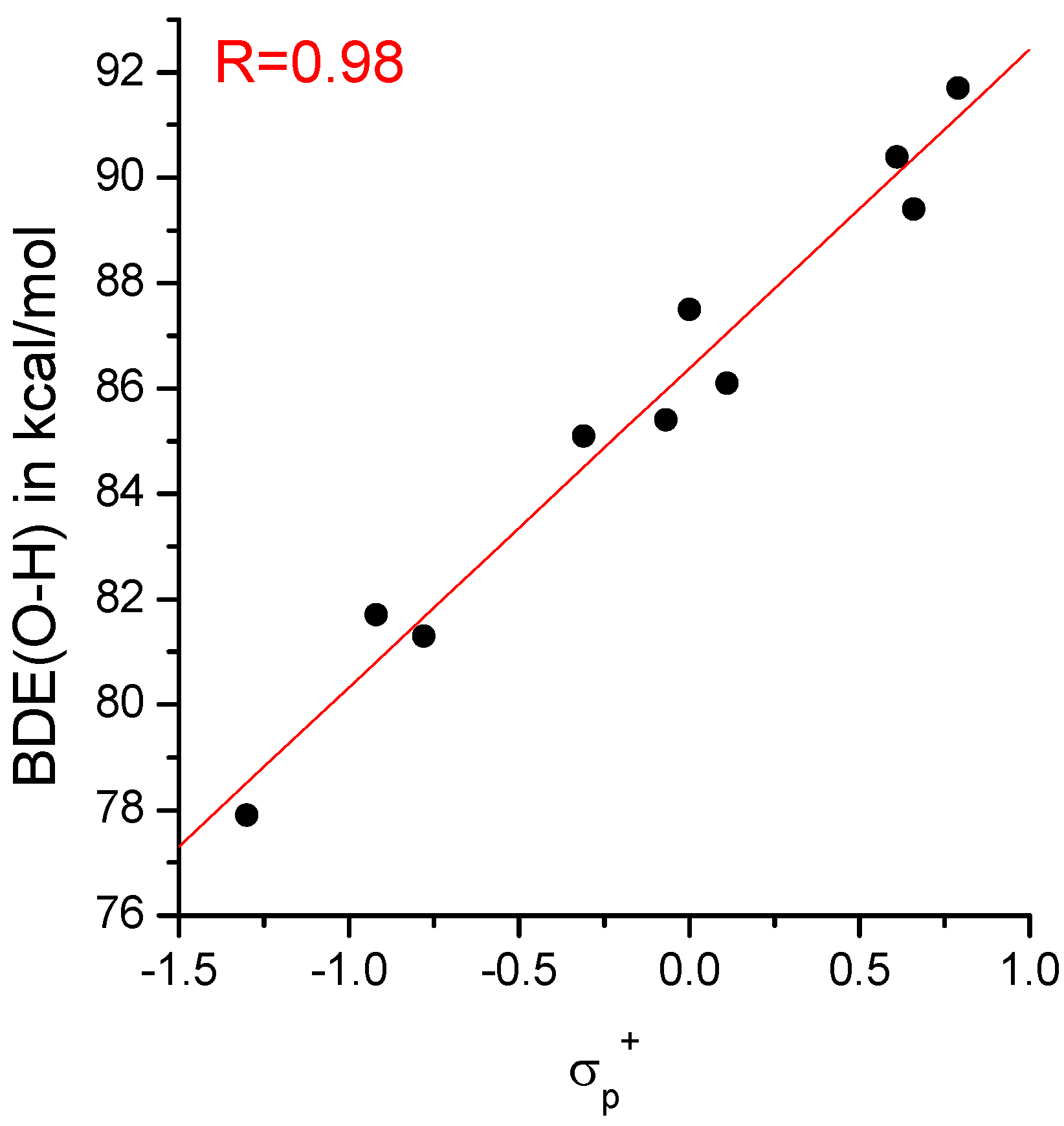
IJMS | Free Full-Text | The O-H Bond Dissociation Energies of Substituted Phenols and Proton Affinities of Substituted Phenoxide Ions: A DFT Study
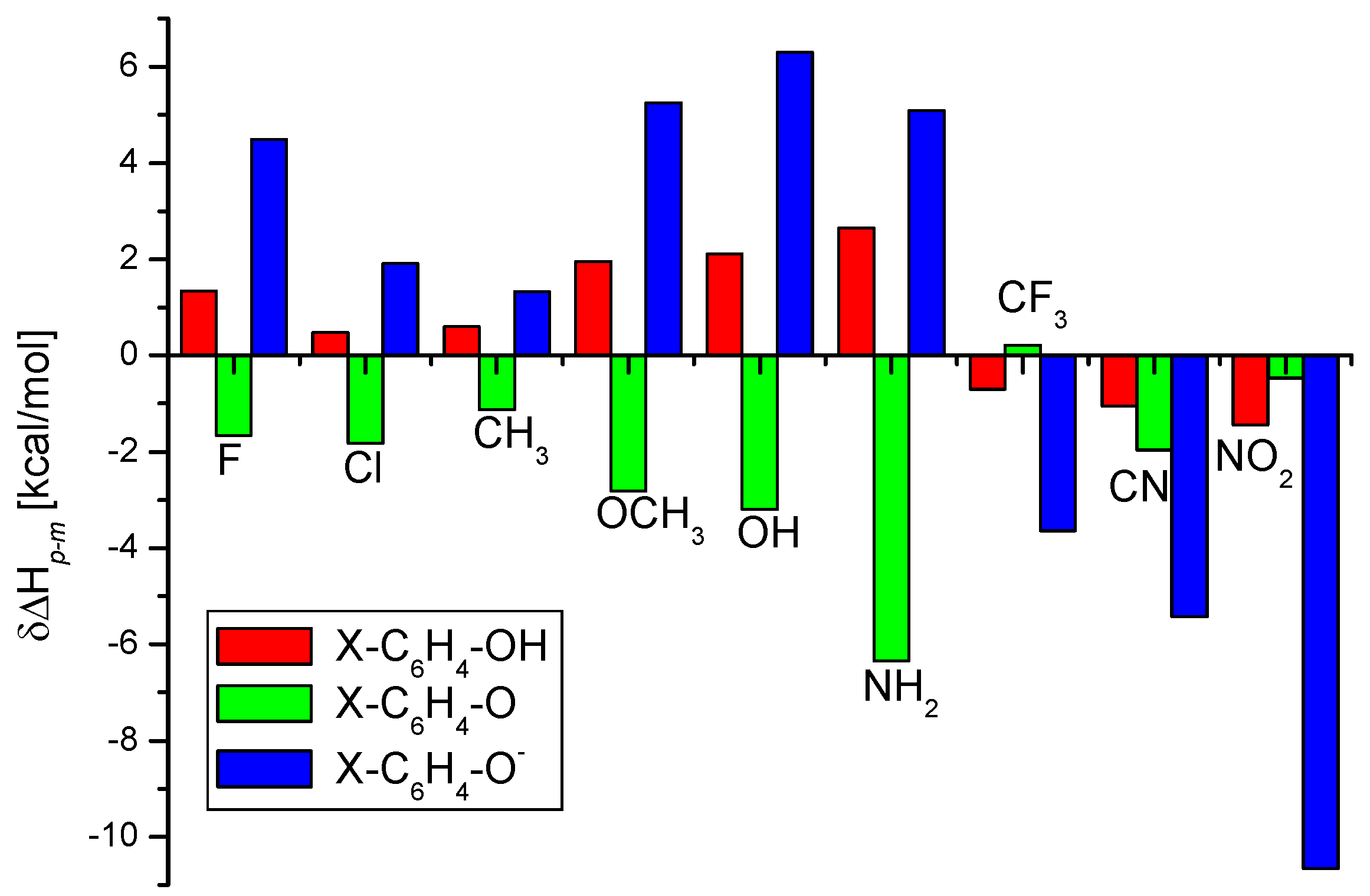
IJMS | Free Full-Text | The O-H Bond Dissociation Energies of Substituted Phenols and Proton Affinities of Substituted Phenoxide Ions: A DFT Study

Exceptionally High O–H Bond Dissociation Free Energy of a Dicopper(II) μ-Hydroxo Complex and Insights into the Geometric and Electronic Structure Origins Thereof | Journal of the American Chemical Society

Benchmark Density Functional Theory Approach for the Calculation of Bond Dissociation Energies of the M–O2 Bond: A Key Step in Water Splitting Reactions | ACS Omega

Dissociating stable nitrogen molecules under mild conditions by cyclic strain engineering | Science Advances

Benchmark study of bond dissociation energy of SiX (XF, Cl, Br, N, O, H and C) bond using density functional theory (DFT) - ScienceDirect

A level Bond Enthalpy (bond dissociation energy) calculations for Enthalpy of Reaction KS5 GCE chemistry revision notes

Study on Chemical Bond Dissociation and the Removal of Oxygen-Containing Functional Groups of Low-Rank Coal during Hydrothermal Carbonization: DFT Calculations | ACS Omega

Density Functional Theory Study of Water Molecule Adsorption on the α-Quartz (001) Surface with and without the Presence of Na+, Mg2+, and Ca2+ | ACS Omega



![PDF] Bond dissociation energies of organic molecules. | Semantic Scholar PDF] Bond dissociation energies of organic molecules. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3aaecb82c2ece7f77647ce525290bf3d9d4eb750/2-Table1-1.png)


![PDF] Bond dissociation energies of organic molecules. | Semantic Scholar PDF] Bond dissociation energies of organic molecules. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3aaecb82c2ece7f77647ce525290bf3d9d4eb750/4-Table2-1.png)